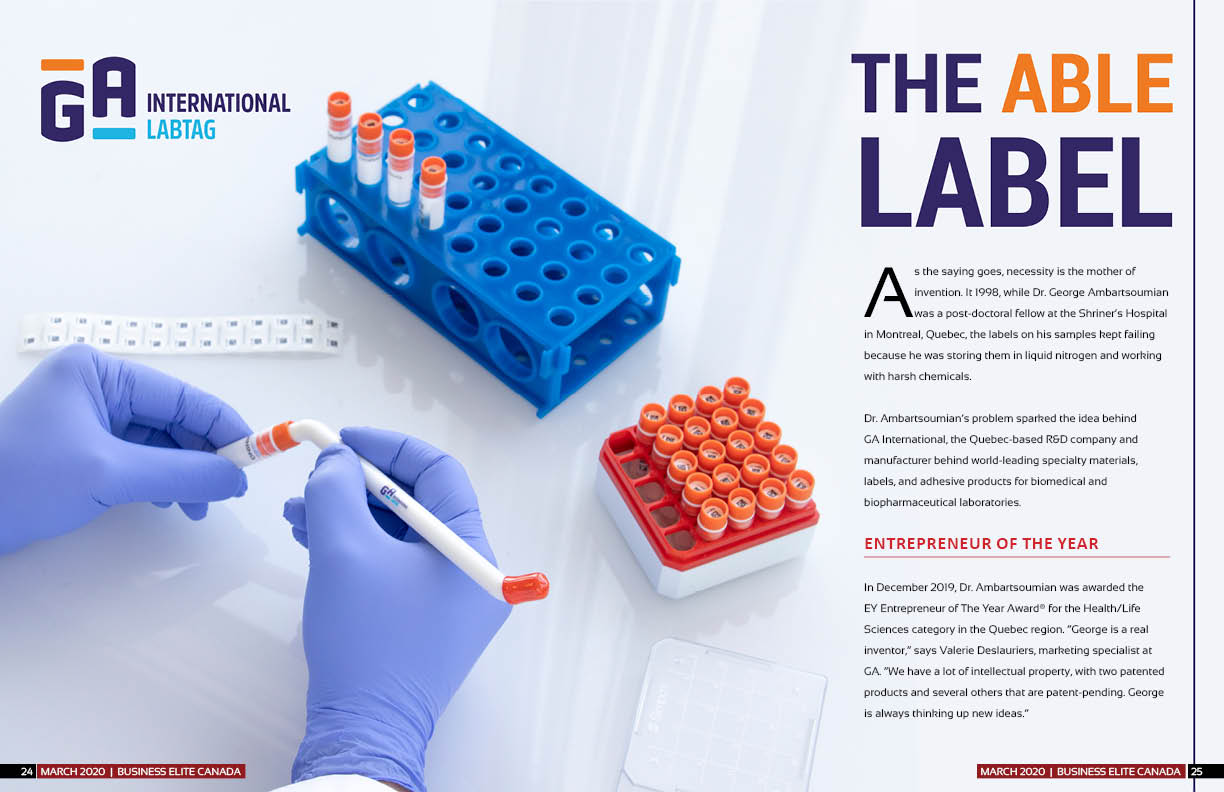
The Able Label
As the saying goes, necessity is the mother of invention. It 1998, while Dr. George Ambartsoumian was a post-doctoral fellow at the Shriner’s Hospital in Montreal, Quebec, the labels on his samples kept failing because he was storing them in liquid nitrogen and working with harsh chemicals.
Dr. Ambartsoumian’s problem sparked the idea behind GA International, the Quebec-based R&D company and manufacturer behind world-leading specialty materials, labels, and adhesive products for biomedical and biopharmaceutical laboratories.
Entrepreneur of the Year
In December 2019, Dr. Ambartsoumian was awarded the EY Entrepreneur of The Year Award® for the Health/Life Sciences category in the Quebec region. “George is a real inventor,” says Valerie Deslauriers, marketing specialist at GA. “We have a lot of intellectual property, with two patented products and several others that are patent-pending. George is always thinking up new ideas.”
Another common thread that weaves through most great success stories is an enthusiastic leader at the helm. “One of the secrets of our company is a happy environment,” says Deslauriers. “His door is always open, and he takes great care of us, bringing in breakfast each week and acknowledging the team’s efforts. Everyone is excited to be productive.”
“There are no formalities here,” smiles Dr. Ambartsoumian.
Demanding Markets
Since its inception in 1999, GA International has been continually designing solutions for the demanding needs of their clients in academic, industrial, and government markets, supplying laboratory identification solutions to biomedical research labs, hospitals, and other healthcare institutions in over 60 countries under its LabTAG™ brand.
Like many great entrepreneurial stories, GA International started out in the founder’s basement, where Dr. Ambartsoumian used a toy label maker to brainstorm his ideas. As GA International’s success grew, so did its offices. Today, GA International comprises a large facility and 65 employees to house its industry-leading R&D capabilities and its ever-expanding roster of exclusive products.
With 90 per cent of its products used in the health care industry, GA International provides labels tailored for extreme conditions, such as long-term cryogenic storage in liquid nitrogen (-196ºC), exposure to harsh solvents, and for other specialty applications requiring exposure to extremely low and high temperatures.
The team strives to make a global impact with specialized identification solutions—constantly growing and expanding by establishing a local presence internationally, adding additional distribution centers to existing and expanding networks.
Quality Control
ISO 9001:2015 certified and ISO 22301:2012 compliant, quality control is of paramount importance for the team who are dedicated to performing rigorous testing on all its labels, says Dr. Ambartsoumian. “Whenever our customers mention our company, they talk about quality and our outstanding scientific and technical support.”
It goes without saying that proper sample identification in the healthcare setting is paramount for patient health and safety. To this end, GA International provides a complete line of data identification equipment, including barcode printers, scanners, as well as data printing and data collection hardware and software.
“We offer the largest selection of cryogenic labels and histology labels worldwide to go with in-depth expertise in labeling and identification solutions for laboratories and clinical trial organizations,” says Dr. Ambartsoumian.
When it comes to biotech or healthcare, the most important thing is data integrity and accuracy of the information. “Our customers always take note that when it comes to clinical data, we’ve never had a single error, something we’re very proud of here,” says Deslauriers. “In clinical trials, labels containing accurate information are critical for success. If duplicate or wrong information is printed, it could cause a major problem for both the patients and the results of the trial. We are always very attentive to making sure these errors don’t occur.”
“Very often, our customers contact us and they need to identify samples for a very specific application or require a label with unique characteristics,” says Dr. Ambartsoumian. “We perform R&D in-house here, producing and delivering small samples of the product as its being designed, so the customer can test it without incurring additional expenses. This facilitates the development of a solution that fits their needs perfectly.”
GA International has “mastered the art” of small batch production without encountering high costs. “Though we do have customers looking for millions of labels, most look for smaller productions. Flexibility is our Number One strength,” says Dr. Ambartsoumian.
“We’re a company of ideas, with new and unique products always in the pipeline. As we continue to expand in the next few years, our future is brighter than ever.”
https://www.ga-international.com/
www.labtag.com